Our research
General Lab Research
Astroglia, also known as astrocytes, are the most abundant glial cells in the mammalian CNS. In addition to their essential functions in maintaining brain homeostasis, they have been recently recognized as essential components in many functional synapses, and they actively modulate various physiological processes in the brain and spinal cord. The mechanisms by which astrocytes acquire their unique morphology and important functions during development remain essentially unknown. The functions of specialized astrocyte subtypes in various CNS physiological conditions also remain largely uncharacterized. Although astrocytes have been clearly implicated in many neurological diseases/disorders as significantly influencing the progression of these diseases, the mechanistic understanding of how astrocytes modulate disease remains limited. This lack of knowledge has become a significant hurdle in understanding the pathological roles of astrocytes in neurologic disorders/diseases.
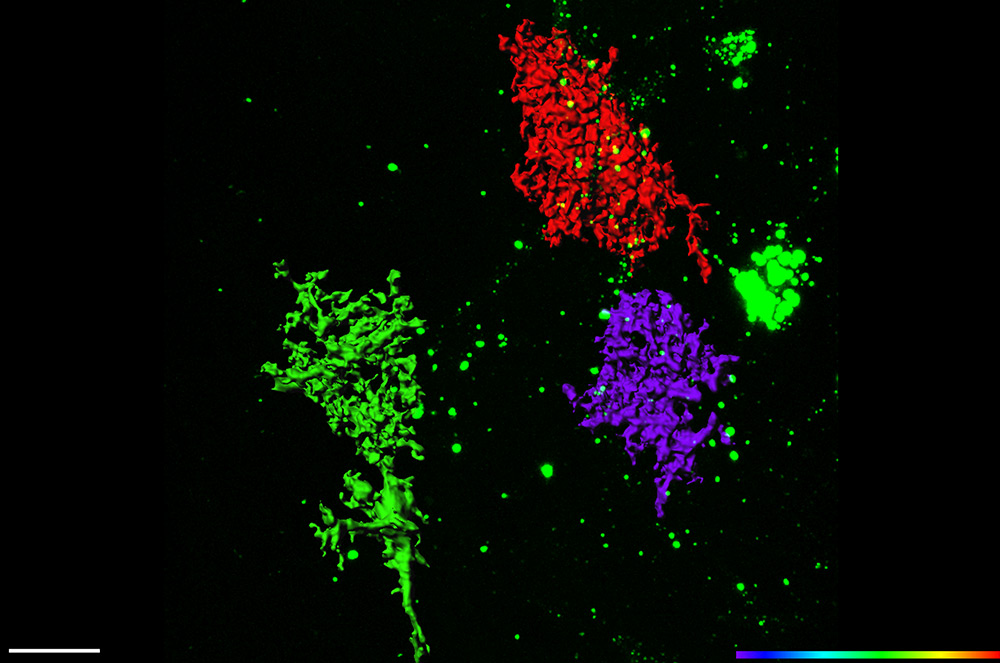
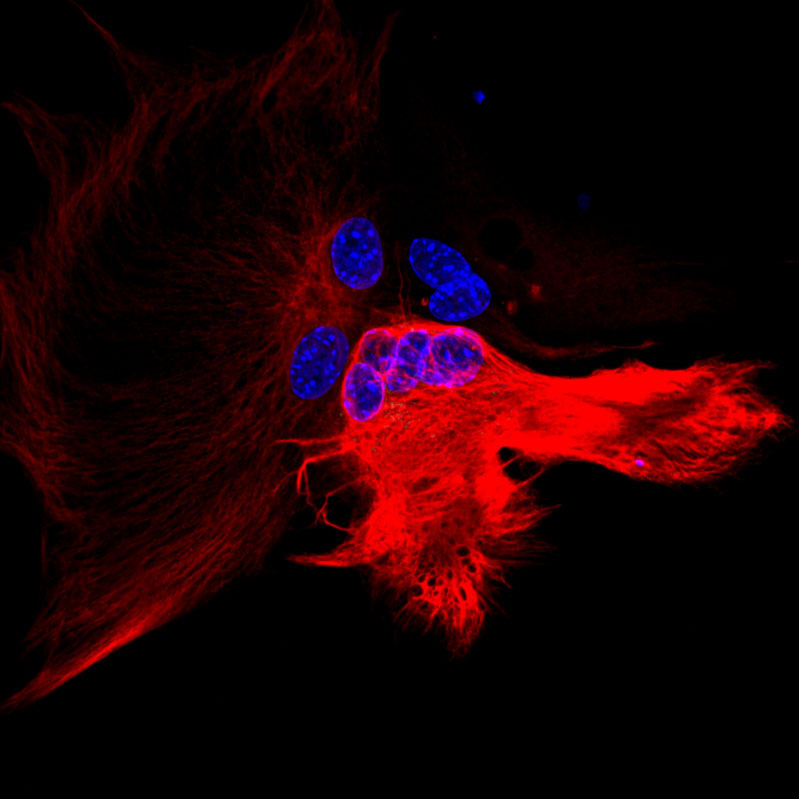
The Yang lab actively explores these areas by specifically investigating how astroglial functions are regulated by neuronal signals (activity and non-activity) during postnatal development and in the adult, how dysregulation of astroglia-mediated glutamate uptake (EAAT2/GLT1) and miRNA biogenesis contributes to synaptic and behavior deficits in FXS, and the molecular and functional heterogeneity of astroglia. Recently, the Yang lab has focused on exosomal signaling in neuron to glial communication by developing new tools, identifying exosomal cargos, and understanding its involvement in Amyotrophic Lateral Sclerosis (ALS) and Alzheimer’s disease (AD) pathogenesis.
Neuron to astroglial communication during development
Despite the importance of astrocytes in the CNS, how they become developmentally mature, especially the acquisition of their unique morphology and dynamic interaction with synapses/vasculatures and induction of astroglial functional genes, remains essentially unknown. We are interested in the molecular mechanisms of astrocytes’ functional maturation during postnatal development, especially the role of neuronal signals in astrocyte maturation. To answer these questions, we employ primary astrocyte and neuron co-cultures and in vivo mouse models in combination with molecular (TRAP-based mRNA profiling), imaging (in vivo astrocyte labeling and time-lapse imaging), genetic (Cre-loxP mouse/viral delivery), and electrophysiological (astrocyte patch and dye-filling) approaches. We have shown that neuronal glutamatergic signals play important roles in the functional maturation of astrocytes during postnatal development (J of Neuroscience, 2014). We have also characterized the regional and cortical-layer-dependent heterogeneity of astrocytes (J of Neuroscience, 2017; Glia, 2019). Recently, we identified and characterized a neuronal exosome-mediated miRNA pathway that regulates astroglial genetic functions (JBC, 2013; Nature Communications, 2019). We are actively investigating mechanisms involved in exosome-mediated neuron to glial communication.
Astroglial mechanisms in Fragile X Syndrome
Fragile X syndrome (FXS) is an inherited neurodevelopmental disorder, caused by the loss of function of FMRP, which closely resembles several autistic syndromes. The functions of FMRP in glial cells remain essentially unknown. In addition, little is known about the role of astroglia in the pathogenesis of FXS. We employ a combination of molecular (TRAP-based mRNA profiling and miRNA), genetic (FMRP conditional knock-out and restoration mice), biochemical, electrophysiological, and behavioral approaches. We have recently demonstrated a unique activation role of FMRP in regulating protein expression in astrocytes. We found that astroglial glutamate transporter subtype GLT1 and glutamate uptake is significantly reduced in the cortex of fragile X mice (Fmr1 KO and cKO mice), contributing to enhanced cortical neuronal excitability (HMG, 2013; J of Neuroscience, 2016). We are continuing our investigation into the role of FMRP in astrocyte development and how astrocytes contribute to the synaptic and behavior phenotypes of FXS.
Exosome signaling in neurodegenerative diseases
Exosomes are a major type of secreted extracellular vesicles that are derived from intraluminal vesicles (ILV) in the early endosomal compartment and are released from multivesicular bodies (MVBs). Recently, exosome-mediated secretion and uptake of disease-causing proteins between CNS cells, including Ab, a-synuclein, tau, misfolded SOD1 and TDP-43, has been reported. These studies support the hypothesis that exosome-mediated secretion and uptake of disease-relevant proteins could underlie propagation of protein aggregates, which is a widely observed pathological feature in neurodegenerative diseases. We have generated a Cre-dependent exosome reporter mouse line that allows selective labeling of exosomes in specific cell types and tracing of their transfer to recipient cells in vivo (Nature Communications, 2019). We are particularly interested in investigating how glial cell secretion (J of Neuroscience, 2014; Exp. Neurol, 2016), including exosomes, affects disease processes. The alteration of exosome signaling in disease and the mechanisms by which exosomes could mediate disease protein propagation is being investigated using biochemical and imaging approaches.
Research Supported by:





